

Robotic surgery has transformed complex surgeries, providing patients with minimally invasive solutions. One such advanced procedure is robotic surgery for mediastinal masses. These masses are located in the mediastinum, the central part of the chest that holds vital organs such as the heart, trachea, and oesophagus. Understanding the process, expectations, and outcomes is essential for patients undergoing robotic surgery for a smoother experience and faster recovery. This blog will walk you through everything you need to know about robotic surgery for mediastinal masses.
Mediastinal masses are abnormal growths within the mediastinum, divided into three compartments: the anterior, middle, and posterior sections. These growths can vary in type and can be benign, malignant, or infectious and often require medical intervention to avoid complications.
When mediastinal masses become symptomatic, causing chest pain, cough, or breathing difficulties, surgical removal is recommended. In recent years, robotic surgery has emerged as a preferred method for performing these complex surgeries with precision.
Robotic surgery suggests several advantages over traditional open surgery or even conventional minimally invasive techniques. It provides surgeons with enhanced visualization, excellent dexterity, and control, making it highly suitable for delicate surgeries within the chest cavity.
This technique ensures that even masses in hard-to-reach areas are addressed effectively, improving patient outcomes.
Proper preparation is important to ensure that the surgery goes smoothly and recovery is efficient. Here are the key steps involved:
1. Medical Evaluation and Diagnostic Tests
2. Consultation with the Surgical Team
3. Fasting and Pre-Surgery Instructions
Robotic surgery for mediastinal masses is typically performed under general anesthesia, suggesting you will be unconscious throughout the procedure. The surgery involves several stages, outlined below:
1. Anesthesia and Patient Positioning
2. Small Incisions and Port Placement
3. Use of Robotic Instruments
4. Mass Removal and Reconstruction
5. Closure of Incisions
Recovery from robotic surgery is generally faster and smoother than traditional open surgery. Here's what you can expect post-surgery:
1. Immediate Postoperative Care
2. Hospital Stay and Monitoring
3. Pain Management and Activity
The recovery period after robotic surgery for mediastinal masses is typically shorter than with conventional surgeries. Here's a timeline of what you can expect:
While robotic surgery is considered safe, it's essential to be aware of potential risks:
Follow-up care is important to ensure complete recovery and monitor for potential complications. During your follow-up appointments:
Robotic surgery for mediastinal masses offers patients a modern, minimally invasive approach that promotes faster recovery, reduced pain, and improved surgical outcomes. With advancements in healthcare technology, robotic-assisted procedures continue to enhance the treatment of complex thoracic conditions.
If you are diagnosed with a mediastinal mass, consulting a healthcare professional about robotic surgery can help you make an informed decision. With proper preparation, careful surgical planning, and comprehensive follow-up care, robotic surgery ensures patients receive the best possible treatment with minimal disruption to their lives

18+ Yrs Exp | 5,700+ Thoracic & Robotic Cancer Surgeries
Dr. Parveen Yadav is a Director and Senior Consultant in Thoracic and Surgical Oncology, specializing in minimally invasive and robotic lung and esophageal surgeries, with advanced training from AIIMS and Tata Memorial Hospital.
View Full Profile Robotic Surgery for Esophageal Cancer in India: Benefits, Recovery and When It Is Recommended
Robotic Surgery for Esophageal Cancer in India: Benefits, Recovery and When It Is Recommended
 Best Bronchopleural Fistula Treatment in Gurgaon: What to Look for in a Thoracic Surgeon
Best Bronchopleural Fistula Treatment in Gurgaon: What to Look for in a Thoracic Surgeon
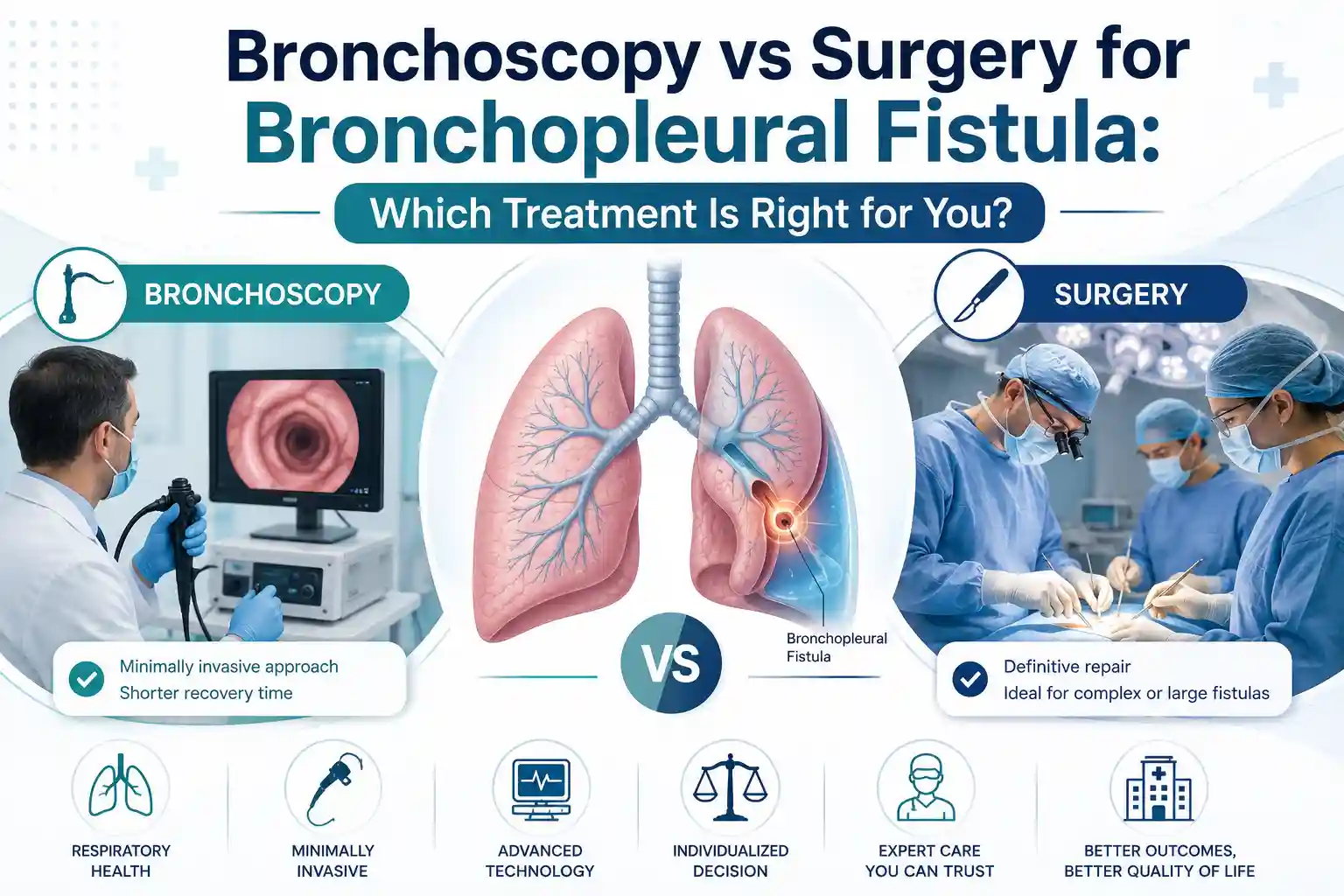 Bronchoscopy vs Surgery for Bronchopleural Fistula: Which Treatment Is Right for You?
Bronchoscopy vs Surgery for Bronchopleural Fistula: Which Treatment Is Right for You?
Learn when robotic surgery for esophageal cancer in India is recommended, its benefits, recovery, and how to consult an expert in Gurgaon.
Choosing the best bronchopleural fistula treatment in Gurgaon? Here is what to look for in a thoracic surgeon, hospital, and care team before you decide.
Bronchoscopy or surgery for bronchopleural fistula? Compare both options, success rates, recovery, and costs. Expert guidance from Dr. Parveen Yadav, Gurgaon.
Copyright 2026 © Dr .Parveen Yadav all rights reserved.
Proudly Scaled by Public Media Solution!