
A 48-year-old man walked into a hospital in Gurgaon last year. He was there for something routine. His orthopedic surgeon wanted a chest X-ray done before a knee replacement. Nothing about his health seemed wrong. No cough. No chest pain. No weight loss. He felt perfectly fine.
But the X-ray told a different story. There was a shadow behind his breastbone. A mass was sitting quietly in his chest, in a space called the anterior mediastinum. Further scans confirmed it was a thymoma, a tumor growing from an organ most adults have completely forgotten about: the thymus gland.
This story is not unusual. According to the American Cancer Society, roughly half of all thymomas are discovered by accident during imaging done for something else. Only about 400 cases are diagnosed each year in the United States alone. It is one of the rarest cancers most people will never hear about... until it shows up on their scan.
As a thoracic surgeon who has treated hundreds of patients with mediastinal tumors, I want to break down everything you need to know about thymoma and thymic carcinoma in plain, simple language. Whether you are a patient, a caregiver, or someone who just received a confusing report, this guide was written for you.
In short: Thymoma and thymic carcinoma are rare cancers that grow from the thymus gland in the chest. They behave very differently from each other, and early treatment can lead to excellent outcomes.
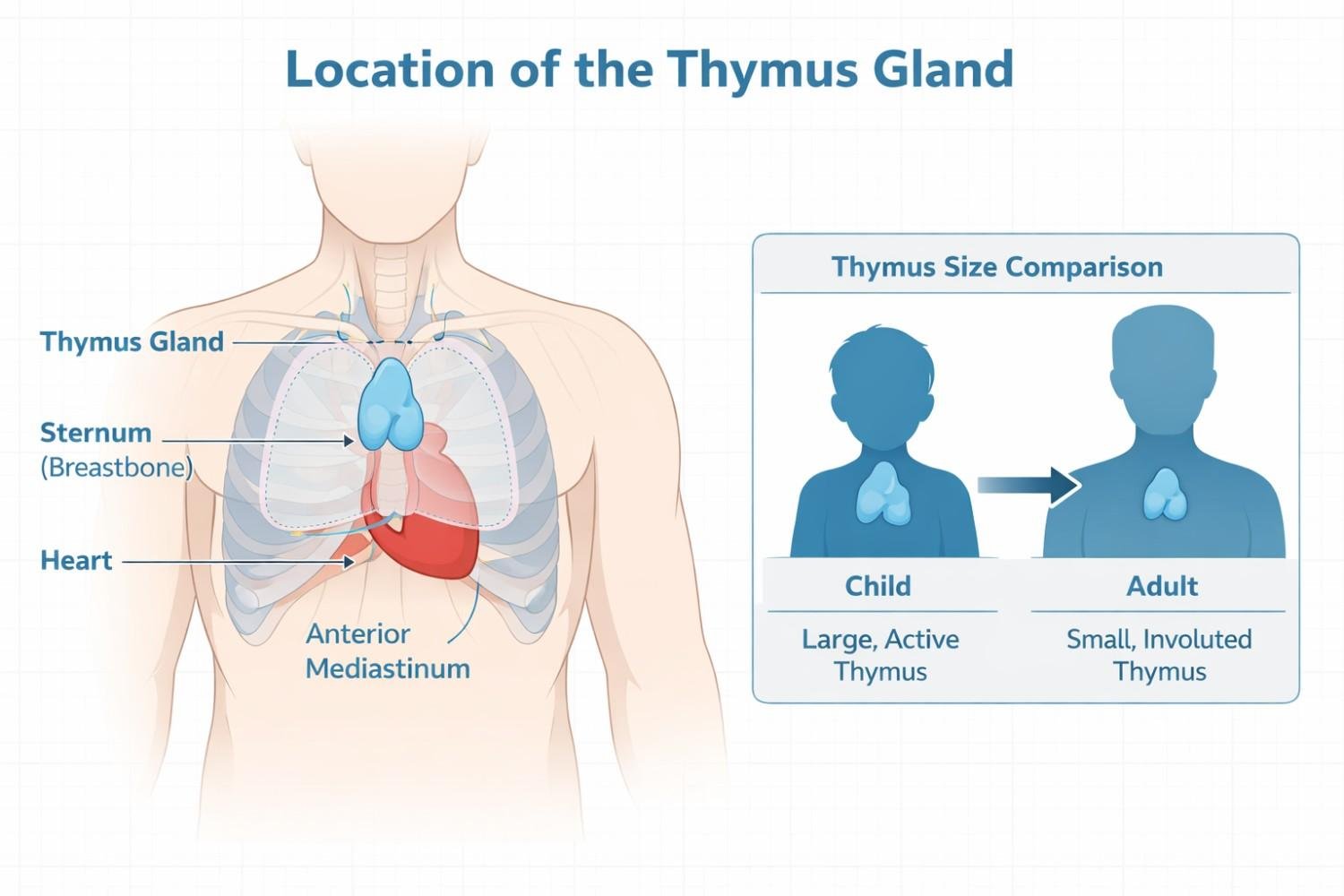
The thymus is a small, butterfly-shaped gland. It sits right behind your breastbone, just above your heart. During childhood, it plays a major role in building your immune system. It trains a type of white blood cell called T-lymphocytes to fight infections.
After puberty, the thymus slowly shrinks. By the time you reach middle age, most of it has turned into fatty tissue. Here is the surprising part: even though the thymus gets smaller with age, tumors in this gland actually become more common as you get older. The peak incidence occurs between ages 70 and 74, according to data from the NCI SEER program. Scientists still do not fully understand why a shrinking organ produces more tumors over time.
The thymus sits in the anterior mediastinum, which is the front part of the space between your lungs. Doctors sometimes call the common tumors found here the "4 Ts": thymoma, teratoma, thyroid tissue, and terrible lymphoma. Among these, thymoma is the most frequent, making up about 50% of anterior mediastinal masses.
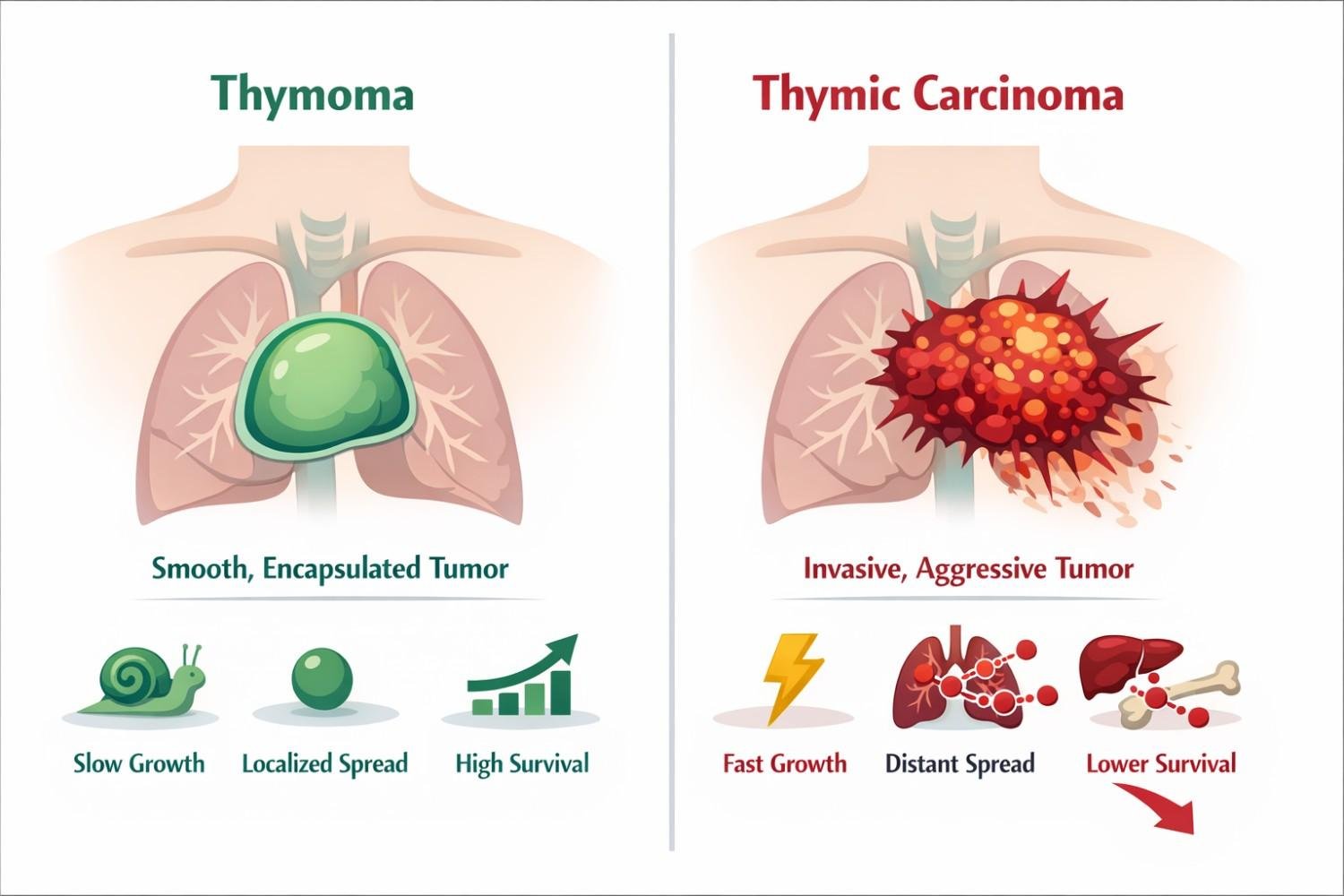
Many people use these terms interchangeably. That is a mistake. While both grow from the same type of cell in the thymus, they behave very differently.
| Feature | Thymoma | Thymic Carcinoma |
| Growth speed | Slow-growing | Fast-growing |
| Spread pattern | Usually stays in or near the thymus | Often spreads to lymph nodes, bones, liver, or lungs |
| Cell appearance | Cells look similar to normal thymus cells | Cells look abnormal and aggressive |
| 5-year survival | 78-90% overall | 30-55% depending on stage |
| Proportion of TETs | ~80% of thymic tumors | ~20% of thymic tumors |
Source: NCI PDQ: Thymoma and Thymic Carcinoma Treatment, 2024
What most people don’t realize is that doctors classify thymomas into multiple subtypes (Type A, AB, B1, B2, and B3) based on how the cells look under a microscope. Types A and AB tend to be the least aggressive. Type B3 sits on the edge and can behave more like thymic carcinoma. This classification, developed by the World Health Organization, directly affects your treatment plan and long-term outlook.
A major update arrived in January 2025 with the 9th edition of the TNM staging system. It introduced a new 5 cm size cutoff for T1 tumors and reclassified how doctors evaluate lung invasion and phrenic nerve involvement. If your staging report was done before 2025, it may use older criteria. This is worth discussing with your surgeon.
Thymoma patients typically show up in one of three ways, according to research published in the Asian Pacific Journal of Cancer Care (2024):
• 30% have symptoms from the tumor pressing on nearby structures, including persistent cough, chest tightness, shortness of breath, or swelling in the face and neck.
• 40% are diagnosed because of myasthenia gravis, an autoimmune condition that causes drooping eyelids, double vision, difficulty swallowing, and muscle weakness.
• 30% have no symptoms at all. The tumor is found by accident on imaging, just like the patient I described at the start.
If you have a persistent cough that lasts more than three weeks, unexplained chest heaviness, or new muscle weakness that gets worse with activity, it is worth seeing a chest specialist. These symptoms deserve proper evaluation, especially in adults over 40.
Most websites will tell you that myasthenia gravis (MG) is linked to thymoma. That is true. MG shows up in 30 to 50% of thymoma patients. But here is the part almost nobody covers: MG is just one of 123 different paraneoplastic syndromes that have been documented with thymoma. A 2019 systematic review in the Journal of Thoracic and Cardiovascular Surgery found that 55% of thymoma patients develop at least one autoimmune condition.
• Good syndrome: A type of immunodeficiency where your body stops making enough antibodies. It leads to repeated infections like pneumonia, sinusitis, and chronic diarrhea. It is often misdiagnosed for years.
• Pure red cell aplasia: The bone marrow stops producing red blood cells properly, causing severe anemia.
• Isaacs’ syndrome (neuromyotonia): Nonstop muscle twitching, cramps, excessive sweating, and stiffness that does not go away with rest.
• Morvan’s syndrome: A rare combination of insomnia, hallucinations, excessive sweating, and muscle twitching. It can be terrifying for patients and families.
• Limbic encephalitis: Memory loss, confusion, seizures, and personality changes caused by the immune system attacking the brain.
Here is another fact that catches many patients off guard: myasthenia gravis can develop after thymectomy, not just before it. A case study in PMC (2023) documented a patient who had zero MG symptoms before surgery, then developed the condition afterward. This is why many experts now recommend testing for MG antibodies before any thymoma surgery, even when the patient feels fine.
The diagnostic process usually starts with a CT scan of the chest. On imaging, thymomas tend to appear smooth and well-defined. Thymic carcinomas look more irregular, with areas of dead tissue, cysts, or calcium deposits.
What surprises many patients is this: unlike most cancers, thymomas often go straight to surgery without a biopsy. If the imaging is clear and the tumor looks resectable, many surgeons skip the needle biopsy entirely. This is standard practice backed by Stanford Medicine and international guidelines.
Other tools in the diagnostic toolkit include:
• MRI, especially useful for checking if the tumor has reached the heart wall or chest wall
• PET scan: thymomas are often PET-negative, while thymic carcinomas light up
• Blood tests to rule out other anterior mediastinal tumors like germ cell tumors (AFP, beta-hCG) or lymphoma (LDH)
• Acetylcholine receptor antibody testing to screen for myasthenia gravis before surgery
Complete surgical removal of the tumor remains the single most important factor in long-term survival. Data from the NCI SEER database shows that for Stage III and IV thymoma, patients who had a complete resection had a 93% five-year survival rate, compared to just 36% for those whose tumors could not be operated on.
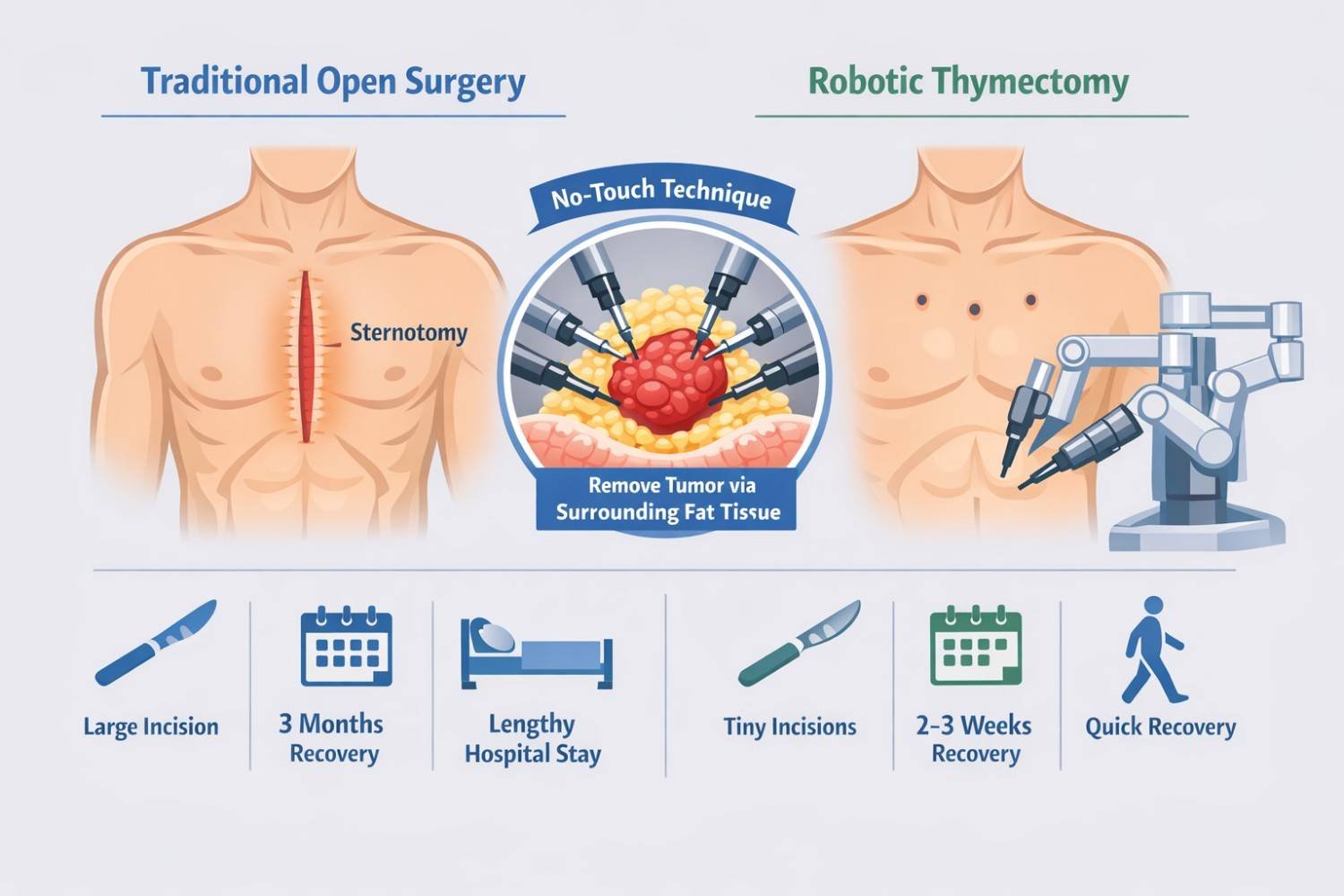
The International Thymic Malignancy Interest Group (ITMIG) recommends that surgeons should never touch the tumor directly during removal. Direct handling can cause cancer cells to spill into surrounding tissue. Robotic surgery, using the Da Vinci XI system, makes this "no-touch" technique far easier to execute than open or traditional methods.
Robotic thymectomy offers several advantages over the traditional open approach:
• Three to four small incisions instead of splitting the breastbone
• 3D magnified vision (10x) for the surgeon
• Wristed instruments that move with more precision than a human hand
• Recovery in 2 to 3 weeks versus 3 months for open surgery
• Equivalent or better cancer outcomes compared to open surgery
For patients seeking advanced mediastinal tumor treatment in Gurgaon, robotic thymectomy at a high-volume center offers the best combination of safety and results. Research published in PMC (2023) confirms that VATS and robotic approaches provide equivalent or better oncological outcomes compared to open surgery.
Radiation therapy is typically used after surgery for Stage II tumors with high-risk features and for Stage III disease. Chemotherapy plays a different role depending on the tumor type: thymomas respond well to cisplatin-based regimens with doxorubicin, while thymic carcinomas are treated with carboplatin and paclitaxel combinations.
The treatment landscape for thymic tumors is evolving fast. Here are the most important recent developments that most websites have not yet covered:
• PECATI Trial (ESMO 2024): The combination of lenvatinib and pembrolizumab showed an 85% one-year survival rate in patients with advanced B3 thymoma and thymic carcinoma. This was presented at the 2024 ESMO Congress and represents a major step forward.
• MARBLE Trial (Lancet Oncology 2025): Atezolizumab combined with carboplatin and paclitaxel showed promising activity in advanced thymic carcinoma.
• Antibody-drug conjugates: Sacituzumab govitecan is now in Phase II trials for thymic tumors, opening up an entirely new class of treatment.
However, there is an important catch. Many thymoma patients have autoimmune conditions that make immunotherapy risky. The same immune system irregularities that cause myasthenia gravis can also trigger severe side effects from checkpoint inhibitors. Researchers at Indiana University and other centers are actively working to understand which patients can safely receive these new treatments.
This might be the most important section of this entire article. Thymoma patients face a significantly higher risk of developing a second, unrelated cancer later in life. A population-based study from the Swedish Cancer Registry (PMC) found these alarming numbers:
• Non-melanoma skin cancer: 10.6 times higher risk than the general population
• Non-Hodgkin lymphoma: 6.8 times higher risk
• Endocrine cancers: 4.7 times higher risk
• Prostate cancer: 3.0 times higher risk
On top of that, thymomas can recur many years, even decades, after the initial treatment. The National Cancer Institute specifically states that lifelong follow-up is needed for thymoma patients. This is not a cancer you treat once and forget about. Annual chest imaging and regular checkups are essential for the rest of your life.
If you have just been diagnosed, here is some encouraging news. Thymoma, when caught early and removed completely, has excellent outcomes.
| Stage (Masaoka) | 5-Year Survival (Thymoma) | Key Factor |
| Stage I (encapsulated) | 90-100% | Surgery alone is usually sufficient |
| Stage II (capsule invasion) | 85-90% | May need radiation after surgery |
| Stage III (organ invasion) | 50-70% | Multimodal treatment needed |
| Stage IV (spread) | 20-50% | Complete resection still improves odds |
Sources: Stanford Medicine; StatPearls/NCBI
A 2024 study published in Scientific Reports analyzed 3,857 patients from the SEER database and found that tumors smaller than 6.5 cm had significantly better survival outcomes. Size matters, and it is another reason why early detection through routine imaging can save lives.
• Thymoma and thymic carcinoma are rare but treatable tumors of the thymus gland in the chest.
• About 50% are found by accident. If yours was, do not panic. Early-stage outcomes are excellent.
• Myasthenia gravis is just one of 123 paraneoplastic conditions linked to thymoma. Ask your doctor to screen you.
• Complete surgical removal is the most important factor in survival. Robotic thymectomy offers the least invasive path to that goal.
• New immunotherapy trials (PECATI, MARBLE) are opening doors for advanced cases.
• Lifelong follow-up is not optional. Second cancers and late recurrence are real risks.
Remember the patient from the start of this article? After a robotic thymectomy, his tumor turned out to be a Stage I thymoma. He was back at work within three weeks. His knee surgery went ahead two months later. Today, he comes in once a year for a follow-up CT scan. That is the power of early detection combined with expert mediastinal tumor treatment in Gurgaon.
If you or a loved one has been diagnosed with a mediastinal mass, a thymoma, or thymic carcinoma, I encourage you to seek a second opinion from a thoracic surgeon who specializes in these rare tumors. At Chest Surgery India, we offer expert evaluation, Da Vinci XI robotic surgery, and comprehensive follow-up care for patients from across India and around the world.
Have you recently been told about a shadow on your chest scan? What questions are on your mind right now?

18+ Yrs Exp | 5,700+ Thoracic & Robotic Cancer Surgeries
Dr. Parveen Yadav is a Director and Senior Consultant in Thoracic and Surgical Oncology, specializing in minimally invasive and robotic lung and esophageal surgeries, with advanced training from AIIMS and Tata Memorial Hospital.
View Full Profile Lung Cancer in Non-Smokers: Symptoms, Causes and When to See a Specialist
Lung Cancer in Non-Smokers: Symptoms, Causes and When to See a Specialist
 Chest Wall Tumor Treatment in Gurgaon: Symptoms, Diagnosis and Best Treatment Options
Chest Wall Tumor Treatment in Gurgaon: Symptoms, Diagnosis and Best Treatment Options
 Early Signs of Bullous Pemphigoid: Itching, Rash and Blisters You Shouldn't Ignore
Early Signs of Bullous Pemphigoid: Itching, Rash and Blisters You Shouldn't Ignore
Know the symptoms, causes, risk factors, and warning signs of lung cancer in non-smokers. Learn when to consult a lung cancer specialist in Gurgaon.
Looking for chest wall tumor treatment in Gurgaon? Learn about symptoms, diagnosis, and best treatment options from an experienced thoracic surgery team.
Learn the early signs of bullous pemphigoid — itching, rash, and blisters. Know when to seek expert bullous disease treatment near you in Delhi & Gurgaon.
Copyright 2026 © Dr .Parveen Yadav all rights reserved.
Proudly Scaled by Public Media Solution!